You are about to leave the SIGNIFOR.com website and enter a website operated by an independent third party. The links to third-party websites contained on SIGNIFOR.com are provided solely for your convenience. Recordati Rare Diseases does not control the opinions, claims or comments contained on any third-party website linked to SIGNIFOR.com, and your activities at those websites will be governed by the policies and practices of those third parties.
SIGNIFOR is the first and only subcutaneously administered,
pituitary-targeted therapy clinically proven to treat Cushing’s disease.

SIGNIFOR® (pasireotide) is similar to somatostatin, a hormone that occurs naturally in the body. It works by reducing the excess release of hormones by targeting the receptors in the tumor most responsible for causing that excess.

SIGNIFOR is given as a self-administered injection, or injection by a caregiver, twice daily.
Cushing’s disease at a glance
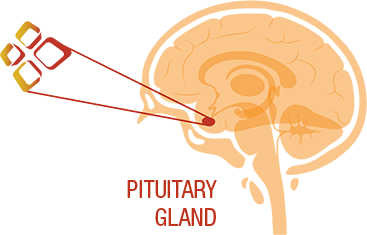
Cushing’s disease is a rare hormonal condition caused when a noncancerous tumor on the pituitary gland produces too much of a natural hormone called adrenocorticotropic hormone (ACTH).
- The extra ACTH then leads to overproduction of another natural hormone called cortisol
- The overproduction of cortisol causes the symptoms of Cushing’s disease
SIGNIFOR works in the pituitary gland to reduce the amount of ACTH released, which in turn helps stop the overproduction of cortisol from the adrenal gland.
Dosing
SIGNIFOR is a twice-daily self-administered
injection, or injection by a caregiver.
What to expect on treatment
While on treatment with SIGNIFOR, improvements in cortisol levels may be experienced.
Cortisol may begin to decrease after starting SIGNIFOR treatment.
TREATMENT WITH SIGNIFOR MAY ALSO LEAD TO REDUCTIONS IN:

Blood pressure

Body mass index

Weight

Waist circumference
In studies, individual patients showed different amounts of improvement in the symptoms of Cushing's disease. Because there was not a group who didn't receive the medication, there were different rates of reductions (lowering) of these symptoms, and blood pressure medications were allowed during this trial, it is not known if these changes were from taking SIGNIFOR or not.
Treatment with SIGNIFOR may lead to:
High blood sugar, also known as hyperglycemia, which may cause excessive thirst, high urine output, increased appetite with weight loss, or tiredness. Your doctor should be monitoring your blood sugar before starting treatment, as well as for the first several weeks and regularly with dose changes.
Gallstones, also known as cholelithiasis, which may cause sudden pain in your abdomen or shoulder area, jaundice (yellowing of the skin or whites of the eyes), nausea, or fever with chills. Your doctor might order an ultrasound to see if this is the cause of these symptoms.
Elevation in liver enzymes, which your doctor may want to test 1-2 weeks after starting treatment, then monthly for 3 months, and eventually every 6 months.
Slow heart rate, also known as bradycardia, or electrical system problem (QT prolongation), both of which may cause weakness, dizziness, or fainting. You should tell your doctor if you have any of these symptoms or if you take any medications for either of these conditions. Your doctor will monitor your heart with an ECG and blood work. Tell your doctor if you have had or have heart problems.
Low cortisol, also known as hypocortisolism, which may cause weakness, fatigue, loss of appetite, nausea, vomiting, low blood pressure, low level of sodium in the blood, or low blood sugar.
If you experience any of these or other side effects, contact your doctor for guidance before discontinuing SIGNIFOR. Learn more about side effects in the Prescribing Information.



